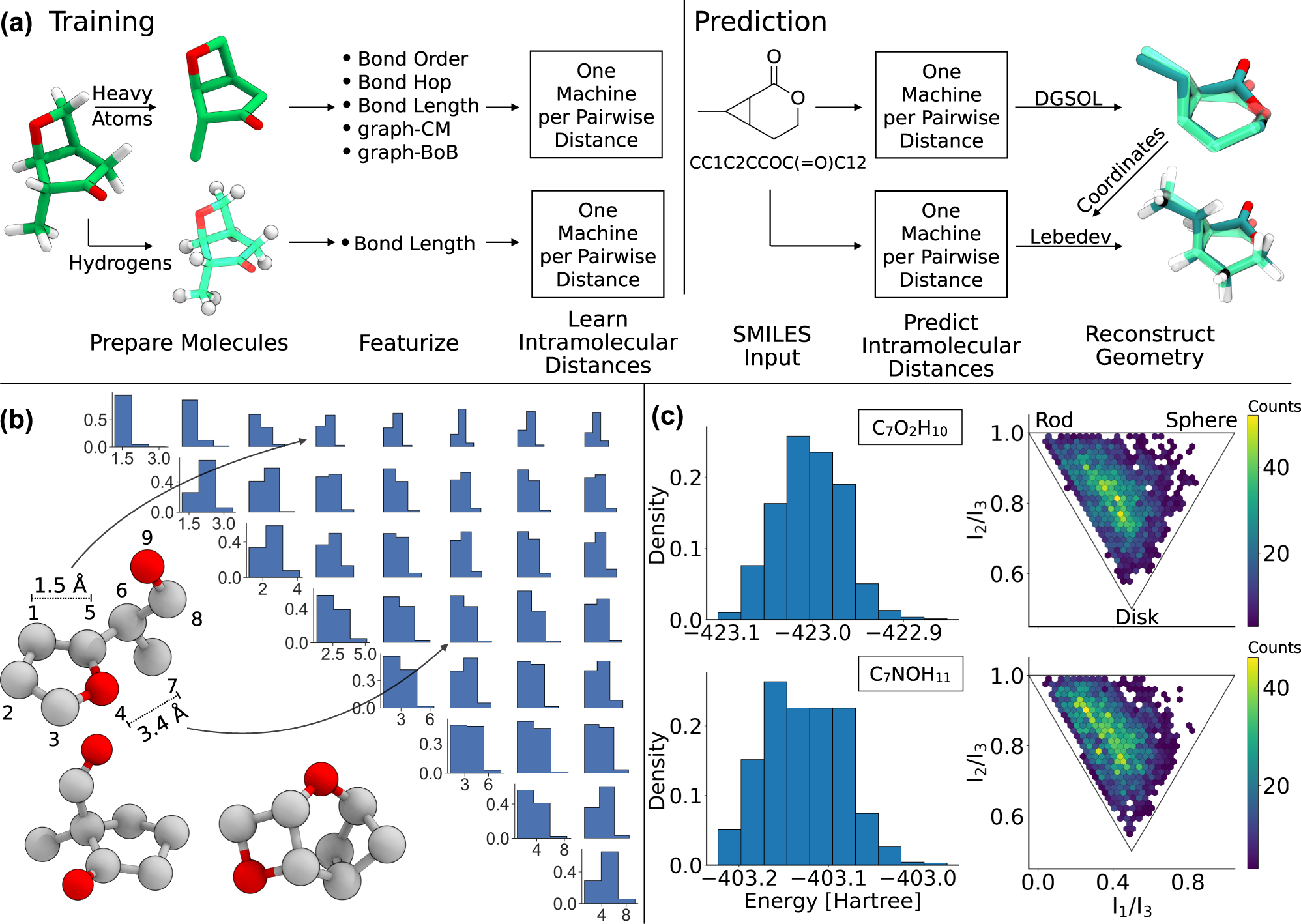
Machine learning based energy-free structure predictions of molecules, transition states, and solids | Nature Communications

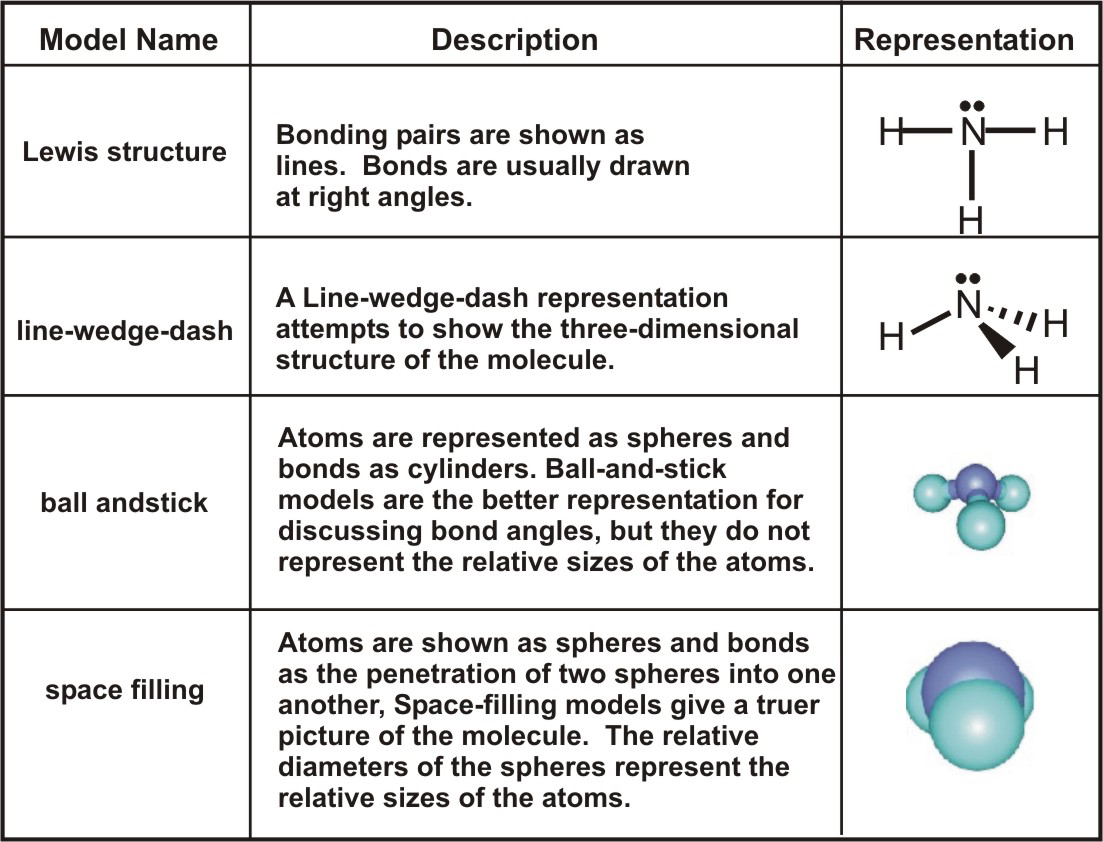
Lewis Dot Structures Lewis Dot structures are used to represent the valence electrons of atoms in covalent molecules Dots are used to represent only the. - ppt download

Simplified S 0 singlet PES at the G3SX//M06-2X/aug-cc-pVTZ level of... | Download Scientific Diagram

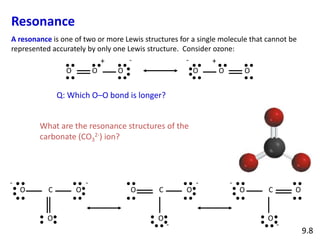
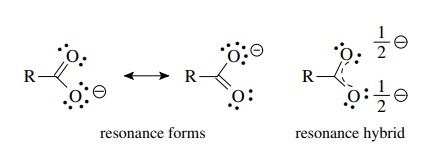
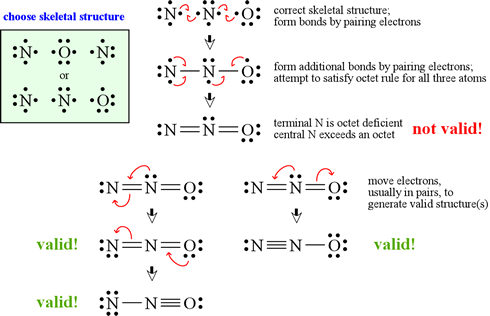
Explain why CO_(3^(2-) ion cannot be represented by a single Lewis structure. How can it be best represented?

Question 44 Explain why CO 32- ion cannot be represented by a single Lewis structure. How can it be best represented?














/ScreenShot2018-11-19at11.40.52PM-5bf3909a46e0fb00510dbd6d.png)
